Data show promising early evidence of anti-tumor activity in patients with relapsed/refractory B cell non-Hodgkin lymphoma
Full data readout of Phase 1 study expected in Q1 2024
Gamida Cell Ltd. (Nasdaq: GMDA), a cell therapy pioneer working to turn cells into powerful therapeutics, today announced new early data in 10 patients with CD20 positive non-Hodgkin lymphoma enrolled in the first three cohorts in an ongoing multicenter Phase 1 study of natural killer (NK) cell therapy candidate GDA-201. The study is designed to evaluate safety and determine the maximum tolerated dose.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20231016887701/en/
(Graphic: Gamida Cell Ltd.)
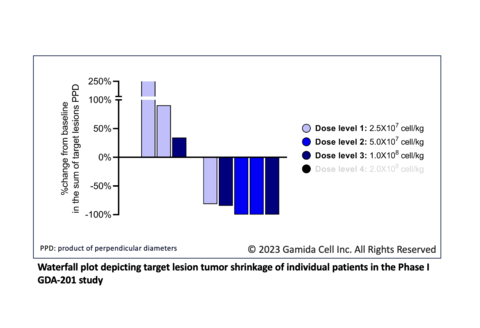
Enrolled patients were heavily pretreated with a median of six prior lines of therapy, including CAR-T cell therapy (six patients) and hematopoietic stem cell transplant (four patients). Preliminary results showed marked shrinkage of target lesions in five patients; efficacy evaluation showed two patients with complete response, two with partial response, and one with stable disease. No dose-limiting toxicities were reported in the 10 patients treated with doses up to 1x108 cells/kg GDA-201 in combination with rituximab.
Activity appears to be dose dependent with two of the three patients in Cohort 3 responding. The fourth and final cohort of the study, at the target dose level of 2x108 cells/kg, is currently enrolling.
“We have demonstrated that our nicotinamide (NAM)-modified NK cells have enhanced metabolic fitness, resistance to oxidative stress and potent cytotoxicity, meaning that GDA-201 has the potential for powerful anti-tumor activity," said Ronit Simantov, MD, Chief Medical and Scientific Officer of Gamida Cell. “We are encouraged by the safety and activity observed thus far in our Phase 1 study of cryopreserved GDA-201, which is consistent with results from the Phase 1 study of a fresh formulation of GDA-201 conducted at the University of Minnesota. We look forward to continuing to follow these patients and completing enrollment in our next cohort, and anticipate sharing the full Phase I data in early 2024.”
The 10 enrolled patients were diagnosed with diffuse large / high grade B cell lymphoma (6), marginal zone lymphoma (2), follicular lymphoma (1) and mantle cell lymphoma (1). Successive cohorts of patients received dose levels of 2.5x107 cells/kg, 5x107 cells/kg and 1x108 cells/kg of GDA-201 with rituximab after fludarabine/cyclophosphamide lymphodepletion. Two patients treated had cytokine release syndrome (grade 1 and grade 2, respectively). The most common grade 3-4 adverse event was transient neutropenia. There were no reported cases of immune effector cell associated neurotoxicity syndrome or graft versus host disease. There was one death from progressive disease.
The NK cells which comprise GDA-201 are powered by Gamida Cell’s proprietary NAM technology, which enhances and expands cells to enhance functionality and phenotype, increase metabolic fitness and reduce oxidative stress. These functional qualities were studied in detail in a recent study published in July 2023 in Science Translational Medicine, which showed that NK cells cultured with NAM had increased energy levels, enhanced ability to arrive at and invade tumors, and an ability to efficiently eradicate malignant cells in the harsh conditions of the tumor microenvironment.
Additionally, the publication includes clinical data from 19 non-Hodgkin lymphoma patients who received the fresh formulation of GDA-201 in a Phase 1 study conducted at the University of Minnesota. GDA-201 exhibited a promising efficacy profile, with an overall response rate of 74% and a complete response rate of 68%. While GDA-201 cells were detected up to 14 days in patients’ blood, the median duration of response was 16 months (range: 5-36 months), suggesting that GDA-201 treatment may prime an endogenous anti-tumor immune response.
About GDA-201
GDA-201 is an intrinsic NK cell therapy candidate being investigated for the treatment of hematologic malignancies. A multicenter Phase 1 study of GDA-201 for the treatment of non-Hodgkin lymphoma is ongoing (NCT05296525). Results are expected in Q1 2024.
GDA-201 is an investigational cell therapy candidate, and its safety and efficacy have not been established by the FDA or any other health authority.
About Gamida Cell
Gamida Cell is a cell therapy pioneer working to turn cells into powerful therapeutics. The company’s proprietary nicotinamide (NAM) technology leverages the properties of NAM to enhance and expand cells, creating allogeneic cell therapy products and candidates that are potentially curative for patients with hematologic malignancies. These include Omisirge™ (omidubicel-onlv), an FDA-approved nicotinamide modified allogeneic hematopoietic progenitor cell therapy, and GDA-201, an intrinsic NK cell therapy candidate being investigated for the treatment of hematologic malignancies. For additional information, please visit www.gamida-cell.com or follow Gamida Cell on LinkedIn, X, Facebook or Instagram.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements as that term is defined in the Private Securities Litigation Reform Act of 1995, including with respect to the Company’s cell therapy candidate, GDA-201. Any statement describing Gamida Cell’s goals, expectations, financial or other projections, intentions or beliefs is a forward-looking statement and should be considered an at-risk statement. Such statements are subject to a number of risks, uncertainties and assumptions including those related to clinical, scientific, regulatory and technical developments and those inherent in the process of developing and commercializing product candidates that are safe and effective for use as human therapeutics. In light of these risks and uncertainties, and other risks and uncertainties that are described in the Risk Factors section and other sections of Gamida Cell’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission (SEC) on August 14, 2023, and other filings that Gamida Cell makes with the SEC from time to time (which are available at www.sec.gov), the events and circumstances discussed in such forward-looking statements may not occur, and Gamida Cell’s actual results could differ materially and adversely from those anticipated or implied thereby. Although Gamida Cell’s forward-looking statements reflect the good faith judgment of its management, these statements are based only on facts and factors currently known by Gamida Cell. As a result, you are cautioned not to rely on these forward-looking statements.
OMISIRGE™ is a trademark of Gamida Cell Inc. © 2023 Gamida Cell Inc. All Rights Reserved.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231016887701/en/
Contacts
Media Contact:
Dan Boyle
Orangefiery
media@orangefiery.com
1-818-209-1692
Investor Contact:
Chuck Padala
LifeSci Advisors
Chuck@lifesciadvisors.com
1-646-627-8390













